Most conventional plastics do not dissolve in water, soil, or the ocean. Their polymer chains are designed to resist these conditions. Whether a plastic dissolves depends on the polymer type, the solvent it contacts, and its structure. Understanding this is key for making smart decisions about plastic disposal, recycling, and material choice.
We often see the misconception that plastic will “break down on its own.” In recycling, this is a persistent and costly belief.
Why Most Plastics Resist Dissolving?
Conventional plastic resists dissolving because of its molecular structure. It consists of long polymer chains with strong carbon-to-carbon bonds. These bonds don’t break in water, soil, or typical environments. They require a compatible solvent, high heat, or UV radiation to weaken.
Water is a polar molecule, but most standard plastics like polyethylene (PE) and polypropylene (PP) are non-polar. This means water cannot interact with their polymer chains on a molecular level. This resistance is a feature, not a flaw. The same chemical stability that makes plastic useful also makes it last for centuries after being thrown away.
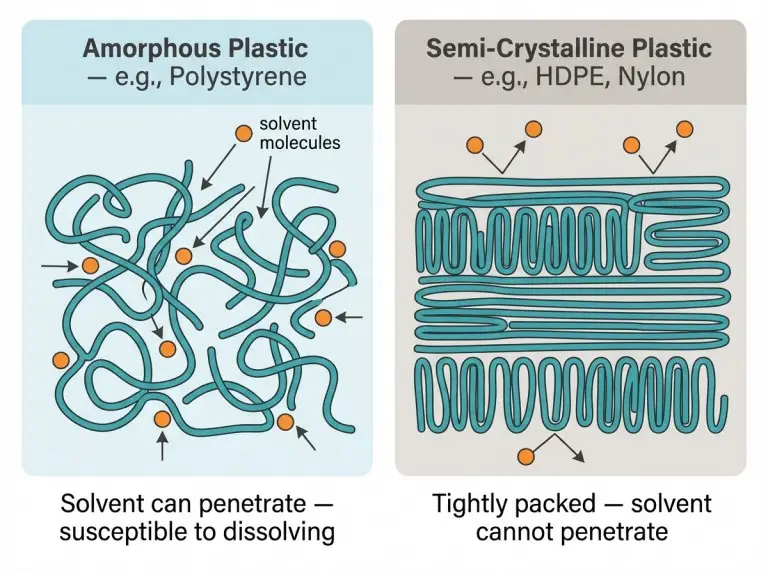
Semi-crystalline plastics like nylon are especially resistant because their tight structure leaves no room for solvents to get in. Amorphous plastics like polystyrene (PS) are more vulnerable to certain organic solvents but still won’t dissolve in water. This guidance applies to petroleum-based plastics; specialty and bio-based materials behave differently.
What Can Actually Dissolve Plastic?
An organic solvent might dissolve a plastic if their polarities are compatible. This is often summarized as “like dissolves like.” Amorphous plastics are more susceptible because their disordered structure lets solvent molecules penetrate the polymer network.
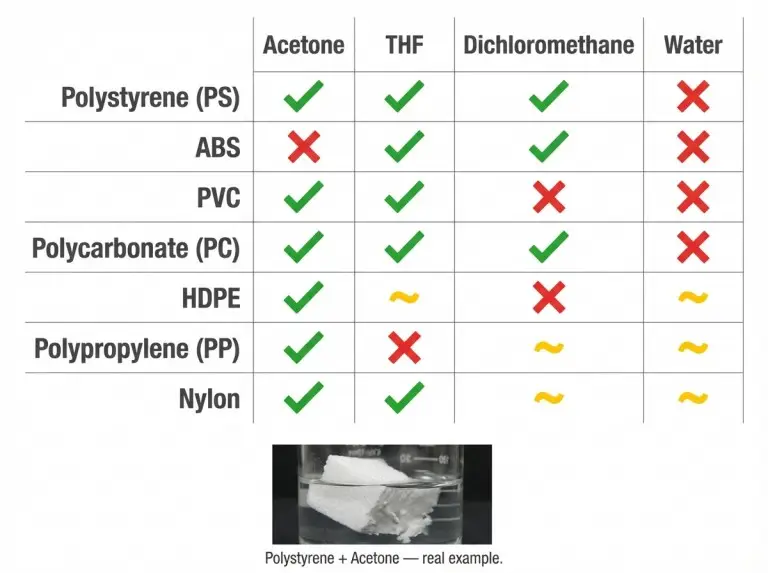
- Tetrahydrofuran (THF) is a broadly effective solvent for many amorphous plastics, including polystyrene, PVC, and polycarbonate.
- Acetone dissolves polystyrene and ABS. This is why Styrofoam collapses when it touches acetone-based products.
- Dichloromethane is used in industrial settings to dissolve polycarbonate and some acrylics.
Highly crystalline plastics like high-density polyethylene (HDPE) and polypropylene resist most common solvents at room temperature. Heat can help overcome this resistance. For example, some polyolefins will dissolve in hot solvents above 120°C, but this isn’t practical outside of industrial settings. The key is the specific polymer type, not just “plastic” in general.
Dissolvable Plastics: A Separate Class
A special category of plastics is engineered to dissolve under specific conditions. These are not your typical plastics. The most common one is polyvinyl alcohol (PVA), which is water-soluble. You can find it in laundry pods, agricultural film, and medical packaging. PVA dissolves because its molecular structure includes hydroxyl groups that interact with water molecules.
The temperature at which PVA dissolves depends on its specific formula. Some grades dissolve in cold water (below 20°C), while others need hot water (above 60°C). This makes them useful for applications that require controlled dissolution.
Bioplastics from corn starch or sugarcane are a related but different category. They are biodegradable, not water-soluble. They break down through microbial activity over months in composting conditions. It’s a common mistake to confuse water-soluble and biodegradable plastics; they have different breakdown methods and disposal needs.

How Conventional Plastic Actually Degrades?
In nature, conventional plastics don’t dissolve; they degrade. The main process is photodegradation. UV radiation from sunlight attacks the polymer chains, breaking them into smaller pieces. Heat and oxygen speed up this process. Most plastics have antioxidants to slow this down, but once they wear out, the plastic becomes brittle.
The result is not dissolution. The plastic doesn’t vanish. Instead, it breaks into tiny pieces called microplastics. These particles, smaller than 5mm, are found in oceans, soil, and even our drinking water. Degradation simply means the plastic mass is spread into the environment in a fragmented form. This distinction is crucial for waste management.
Chemical Recycling via Dissolution
Controlled plastic dissolution is important in an emerging industrial process: chemical recycling. This method uses specific solvents to dissolve target polymers from mixed waste. After separation from contaminants, the polymers are precipitated out as purified material for reuse. This process isolates and recovers plastics without breaking down the polymer chains.
Another path, called depolymerization, uses heat and chemicals to break polymers down into their basic monomer building blocks. Pyrolysis is another chemical recycling method that converts mixed plastic waste into fuel using heat without oxygen. These routes are useful for mixed or contaminated plastics that cannot be mechanically recycled.
Why Mechanical Processing Is Still Key
For most plastic waste, mechanical size reduction is the most cost-effective first step in recycling. Plastic crusher machines turn bulk waste into uniform granules or flakes. These can be re-processed directly or prepared for other recycling methods.
Efficiency in mechanical recycling depends on particle size consistency and contamination control. When material is properly sorted and processed, the resulting recyclate keeps its quality for reuse. If different resin types are mixed, the quality drops, and processors may reject the material. Matching the crusher to the input material is the most important factor for a successful recycling operation.
Conclusion
Most plastics are designed to resist dissolving in water and other common substances. Whether a specific plastic can be dissolved depends on its chemical structure and the solvent used. Dissolvable plastics like PVA are an engineered category for specific uses.
At IPG, we help recyclers and manufacturers improve their plastic waste processing. We find that assuming plastic will disappear on its own leads to contamination and higher costs. A properly specified crushing system addresses these problems at the source. The best recycling operations begin with clear resin identification and matched equipment.
If you are looking for plastic crushing equipment, share your material types and goals with our team. We can help you find the right crusher configuration for your needs.
FAQ
Does plastic dissolve in water?
Most plastics do not. They are non-polar, while water is polar, making them chemically incompatible. The main exception is PVA (polyvinyl alcohol), which is designed to be water-soluble.
What liquid will dissolve plastic?
It depends on the plastic. THF dissolves polystyrene and PVC. Acetone dissolves polystyrene and ABS. Crystalline plastics like HDPE resist most common solvents at room temperature.
Does plastic dissolve in the ocean?
No. Plastic in the ocean degrades from sunlight and wave action into smaller fragments, forming microplastics. The polymer mass remains in the environment.
How long does it take for plastic to break down?
It depends on the plastic type and environment. Estimates range from 20 years for a thin film to 500 years for rigid plastics. This is a degradation timeline, not dissolution.
What is dissolvable plastic made of?
Most are made from polyvinyl alcohol (PVA) or starch-based biopolymers. PVA dissolves in water, while starch-based plastics are broken down by microbes.
Can plastic be recycled instead of dissolved?
Yes. For most plastic waste, mechanical recycling using crusher machines is the main route. Chemical recycling is a complementary option for mixed or contaminated plastics.


